Efficacy


Primary analysis: UNPRECEDENTED OS benefit with 1L KIMMTRAK2-8
Largest phase 3 trial in metastatic uveal melanoma (mUM) evaluated first-line treatment with KIMMTRAK vs investigator’s choice (IC)2,9
- Investigator’s choice included checkpoint inhibitors (pembrolizumab and ipilimumab) or chemotherapy (dacarbazine)2,9
First-line treatment with KIMMTRAK significantly extended median overall survival (mOS) by 6 months vs IC2,9


≈50% reduction in the risk of death2,9
At the time of the data cutoff for the first interim analysis, median duration of follow-up was 14.1 months.2
HISTORIC 5-YEAR follow-up analysis of OS10
KIMMTRAK doubled the likelihood of being alive at 5-years: 16% vs (8%) with investigator's choice10,†

Patients assigned to KIMMTRAK showed a long-term survival advantage at 5 years (KIMMTRAK median overall survival 21.6 months [19.0-24.3] vs 16.9 months [12.9-19.5] with investigator’s choice, 95% CI).10
As of July 2, 2025, all study participants had the opportunity for at least 60 months of follow-up, with a median follow-up of 62.4 months.10
Of the patients surviving at least 5 years10,11:
- 44% (11 of 25) in the KIMMTRAK arm reported ONLY receiving KIMMTRAK
- 86% (6 of 7) in the control group received subsequent KIMMTRAK
The planned exploratory 5-YEAR overall survival analysis occurred after the protocol-specified final analysis and was not tested for statistical significance. No statistical conclusions can be drawn.
CI, confidence interval; HR, hazard ratio; OS, overall survival.
- *Based on Komodo US medical claims from February 2022-December 2025 and calibrated with actual vials sold.1
- †Investigator’s choice included checkpoint inhibitors (pembrolizumab and ipilimumab) or chemotherapy (dacarbazine).10
The OS benefit observed with KIMMTRAK was only in the 1L setting.10
Pivotal trial
Select secondary endpoints
- Progression-free survival (PFS) and objective response rate (ORR) were evaluated using Response Evaluation Criteria in Solid Tumors (RECIST) version 12,9
- A statistically significant PFS benefit was seen with KIMMTRAK compared to investigator’s choice* (3.3 months vs 2.9 months respectively, HR = 0.73; 95% CI, 0.58-0.94; P = 0.0139)2,9
At the time of the data cutoff for this first interim analysis, median duration of follow-up was 14.1 months.2

3-YEAR follow-up analysis
Time to objective response
2.9 months with KIMMTRAK
(range, 1.2 to 22.2) vs 4.1 months with
IC* (range, 2.0 to 11.8).12
Median duration of response
11.1 months with KIMMTRAK vs 9.7
months with IC.12,*
- In the 3-YEAR follow-up analysis, only ORR (11%) and SD (35%) with KIMMTRAK changed12
- Select secondary endpoints remained consistent at the 5-YEAR follow-up analysis10
SD, DCR, time to objective response, and duration of response data are not includedin the Prescribing Information. The statistical significance of SD, DCR, and time to objective response data is not known, and data should be interpreted with caution.
- * Investigator’s choice: checkpoint inhibitors (pembrolizumab and ipilimumab) or chemotherapy (dacarbazine).2,9,10,12
- a Not formally tested.2
- b DCR was defined as a complete response (CR) + partial response (PR) + SD.2,13
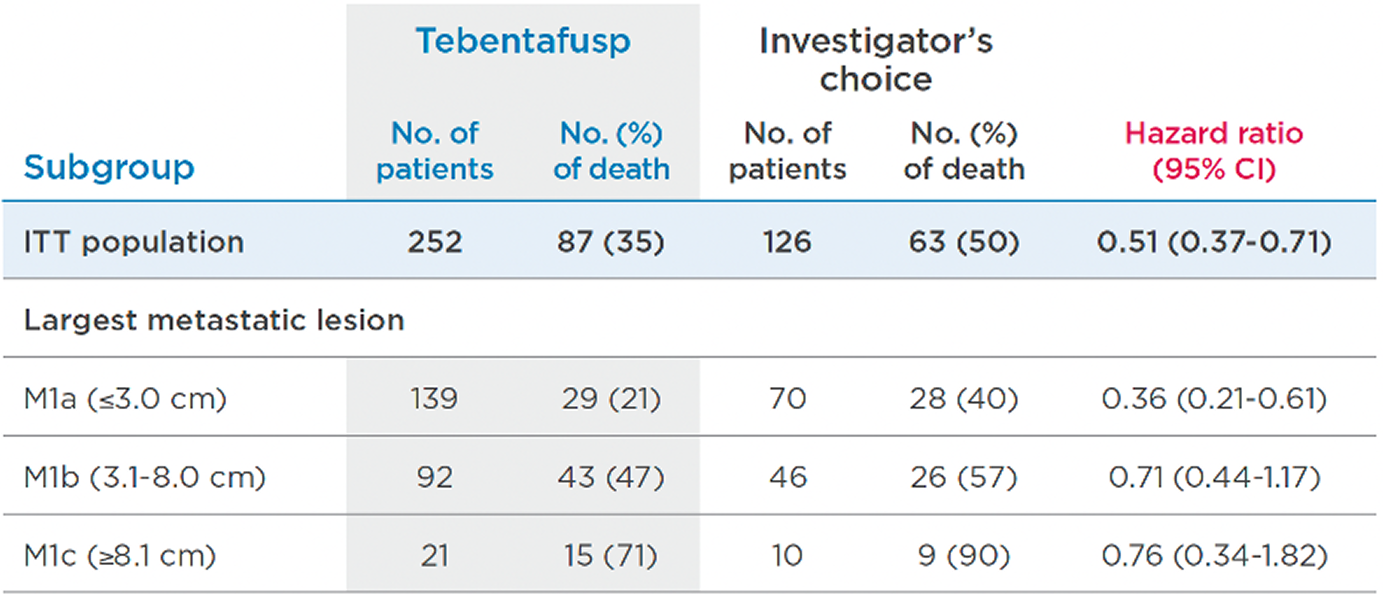
Subgroup analyses of OS
KIMMTRAK was favored numerically in the subgroup analyses of OS per largest metastatic lesion10,13
PRIMARY, PRESPECIFIED13

5-YEAR FOLLOW-UP, PRESPECIFIED10
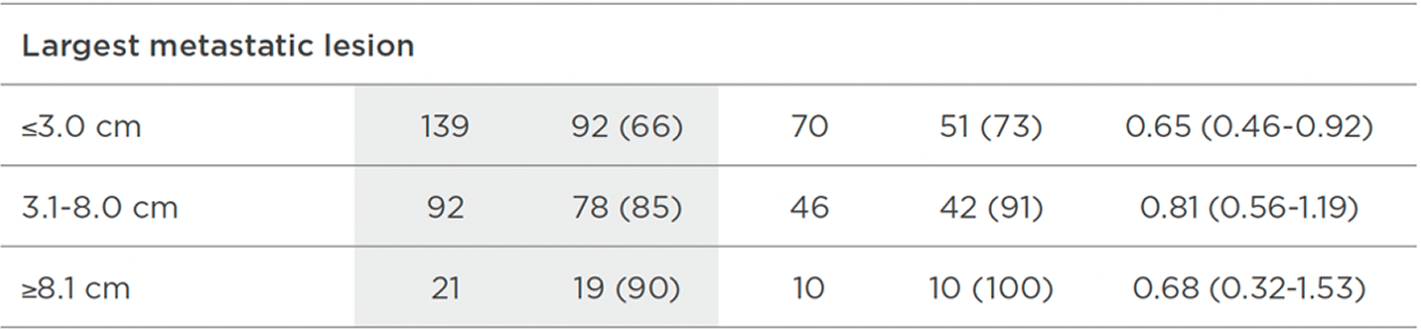

The prespecified patient subgroup analyses were not powered to detect treatment effect differences between subgroups. Data are for informational purposes only.
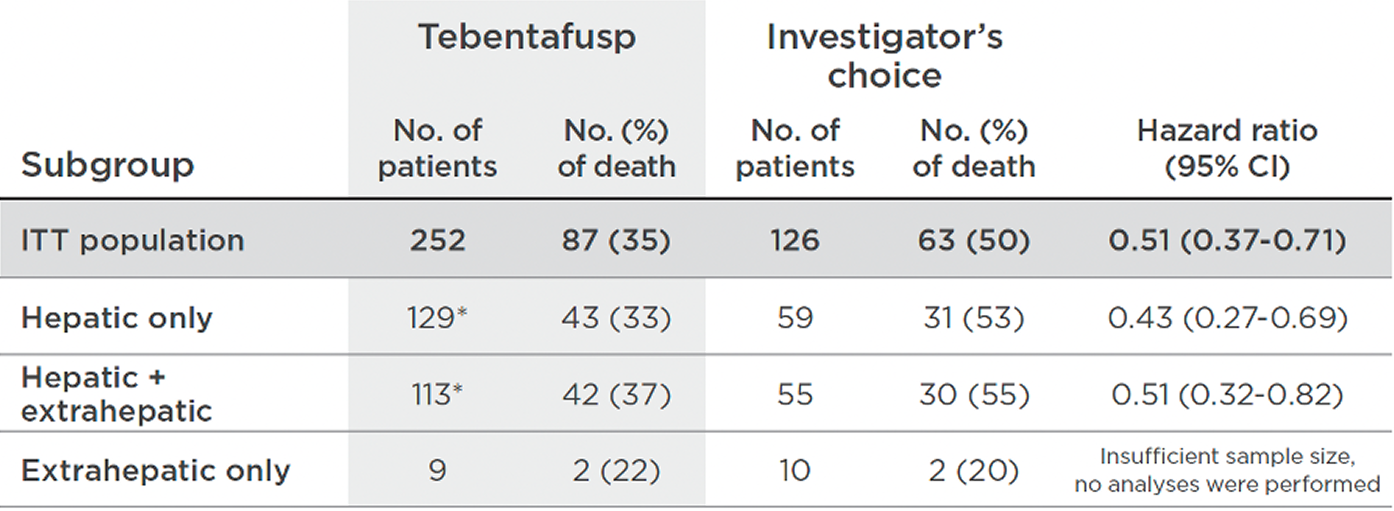
In the subgroup analyses of OS per metastatic disease location, KIMMTRAK was favored numerically in hepatic and hepatic plus extrahepatic patients10,14
PRIMARY, POST HOC14

5-YEAR FOLLOW-UP, POST HOC10
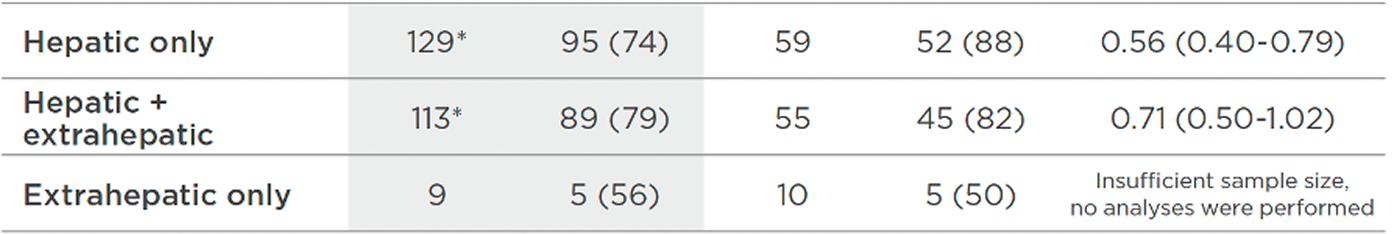

The updated analyses with additional 5-years of follow-up are provided only as descriptive clinical information, and no conclusions can be drawn.
Subgroup analyses were exploratory in nature. This study was not powered to detect differences between treatments based on subgroups. Results of the post hoc analyses are descriptive, are not controlled for type 1 error, and should be interpreted with caution.
CI, confidence interval; ITT, intention-to-treat.
* Post 2021 NEJM publication (Nathan et al.), 2 patients were reclassified from hepatic only to hepatic plus extrahepatic disease.14
Additional analysis
5-YEAR follow-up analysis
Post hoc analysis of patients with a best overall response of progressive disease (PD) by day 100

The post hoc overall survival among patients with best overall response of disease progression analysis by day 100 after randomization occurred after the protocol-specified final analysis and was not tested for statistical significance. These data are not included in the Prescribing Information. The statistical significance is not known and data should be interpreted with caution.
Patients who continued treatment after initial radiographic progression experienced longer post-progression survival compared to those who stopped treatment.10
Treatment with KIMMTRAK should be continued while patient is deriving clinical benefit and in the absence of unacceptable toxicities.9
- Median follow-up: 70.8 months (95% CI, 47.4-Not Calculated (NC))10
- Over half (57%; n = 139) of patients who received KIMMTRAK were treated beyond initial radiographic progression vs 25% (n = 28) in the control group12
- *Investigator’s choice: checkpoint inhibitors (pembrolizumab and ipilimumab) or chemotherapy (dacarbazine).10
Indication
Important Safety Information Including Boxed Warning
KIMMTRAK is indicated for the treatment of HLA-A*02:01-positive adult patients with unresectable or metastatic uveal melanoma.
WARNING: CYTOKINE RELEASE SYNDROME
Cytokine Release Syndrome (CRS), which may be serious or life-threatening, occurred in patients receiving KIMMTRAK. Monitor for at least 16 hours following first three infusions and then as clinically indicated.
Indication and Important Safety Information Including Boxed Warning
Indication
KIMMTRAK is a bispecific gp100 peptide-HLA-directed CD3 T cell engager indicated for the treatment of HLA-A*02:01-positive adult patients with unresectable or metastatic uveal melanoma.
Important Safety Information Including Boxed Warning
WARNING: CYTOKINE RELEASE SYNDROME
Cytokine Release Syndrome (CRS), which may be serious or life-threatening, occurred in patients receiving KIMMTRAK. Monitor for at least 16 hours following first three infusions and then as clinically indicated. Manifestations of CRS may include fever, hypotension, hypoxia, chills, nausea, vomiting, rash, elevated transaminases, fatigue, and headache. CRS occurred in 89% of patients who received KIMMTRAK with 0.8% being grade 3 or 4. Ensure immediate access to medications and resuscitative equipment to manage CRS. Ensure patients are euvolemic prior to initiating the infusions. Closely monitor patients for signs or symptoms of CRS following infusions of KIMMTRAK. Monitor fluid status, vital signs, and oxygenation level and provide appropriate therapy. Withhold or discontinue KIMMTRAK depending on persistence and severity of CRS.
Skin ReactionsSkin reactions, including rash, pruritus, and cutaneous edema occurred in 91% of patients treated with KIMMTRAK. Monitor patients for skin reactions. If skin reactions occur, treat with antihistamine and topical or systemic steroids based on persistence and severity of symptoms. Withhold or permanently discontinue KIMMTRAK depending on the severity of skin reactions.
Elevated Liver Enzymes Elevations in liver enzymes occurred in 65% of patients treated with KIMMTRAK. Monitor alanine aminotransferase (ALT), aspartate aminotransferase (AST), and total blood bilirubin prior to the start of and during treatment with KIMMTRAK. Withhold KIMMTRAK according to severity.
Embryo-Fetal ToxicityKIMMTRAK may cause fetal harm. Advise pregnant patients of potential risk to the fetus and patients of reproductive potential to use effective contraception during treatment with KIMMTRAK and 1 week after the last dose.
The most common adverse reactions (≥30%) in patients who received KIMMTRAK were cytokine release syndrome, rash, pyrexia, pruritus, fatigue, nausea, chills, abdominal pain, edema, hypotension, dry skin, headache, and vomiting. The most common (≥50%) laboratory abnormalities were decreased lymphocyte count, increased creatinine, increased glucose, increased AST, increased ALT, decreased hemoglobin, and decreased phosphate.
Please see full Prescribing Information, including BOXED WARNING for CRS.


